Data for Life: Developments in Data and Privacy for Life Sciences (Guest Blog)
It has been over five years since I worked on the Salford Lung Study data platform for real world evidence. The experience of supporting and developing systems to support real world trials left a big impact on me: I knew we were first to achieve certain goals in real world evidence but I knew we were also creating our own standards due to the lack of developed standards. Now in 2023 I believe that globally we have several standards which have emerged that will expedite research and citizen confidence in participation in research.
OMOP (The Observational Medical Outcomes Partnership)
The emergence of OMOP (https://www.ohdsi.org/data-standardization/) as an open data set for recording the healthcare observations for research is excellent news for research globally. It is backed by the FDA, multiple industry partners and has seen global adoption. This standardised data set and vocabulary allows truly global research to occur with patient observations being recorded in a standardised way.
Below I have indicated how this standard sits with the other global standards used in Healthcare systems.
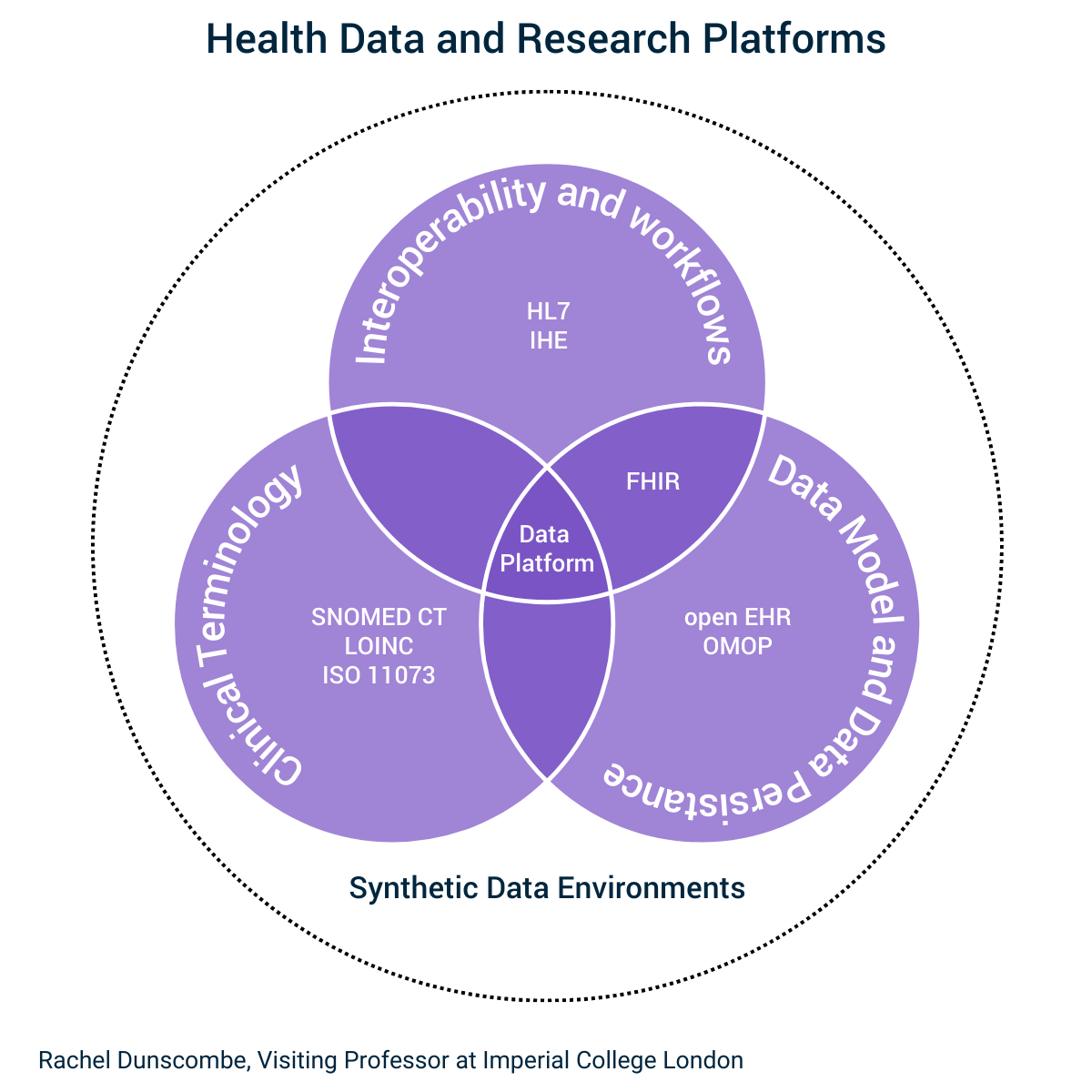
(With thanks to EY and Aloha McBride for allowing me to first publish this)
Healthcare systems, research entitles and life science companies will all need to build OMOP capabilities into their systems to support future research.
PROMS (Patient Recorded Outcomes)
PROMS as a concept has been talked about for many years but emerging global standardisation unlike OMOP has not fully emerged. PROMS is however an essential tool to achieve a full 360 of the patient experience and outcomes in research. The leading standards body for PROMS is ICHOM (https://www.ichom.org/ ) who have for many years been creating global standards with clinical and health system representation across the globe.
Despite the emergence of such standards we still see health system players defining and creating their own patient reported outcomes. Without a doubt data from our citizens is required to enhance the safety and speed of research a question for us today is do we back ICHOM or one of the other emerging standards such as UK based Open Outcomes https://apperta.org/openOutcomes/ . However this may emerge we are professionals have a job to ensure we do not allow a proliferation of standards which slow research progress.
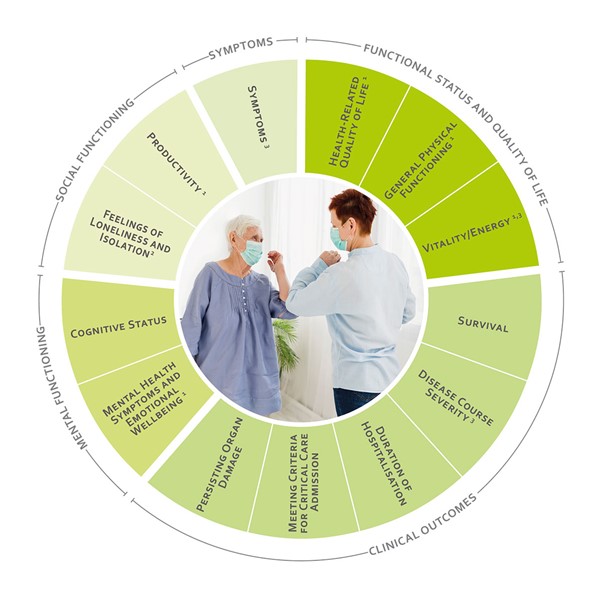
Example of ICHOM Covid-19 PROMS (copywrite ICHOM)
Synthetic Data
The holy grail of research is to be able to work with patient data and data sets without concerns for individual privacy. Imagine the ability to do undertake a population review to see if a population would support a study or evaluate a city to see if there would be enough candidates in a disease group without causing privacy issues. What could we achieve if we could determine correlation between factors for diseases in real population groups?
In the last two years this has become a reality with the emergence of clinically aligned synthetic data tools. These tools take the attributes of a population and create a new data set for research to allow examination of the attributes of cohorts without records of real individuals being involved.
This concept is now being used in the US and Israel as well as in the UK. A fantastic example of how this can improve research and patient outcomes is the UK’s own Breathe hub (https://www.breathedatahub.com/ ) a collaboration between universities, the NHS and life sciences. Here synthesised data is being used to expedite insights and research into respiratory conditions in order to improve speed of life sciences and healthcare system innovation.
Future view
In order for us in the UK to progress as a life sciences centre of excellence we need to embrace and participate in standards and methods that improve our ability to gain data and gain insights. I would encourage you to explore the above concepts and certainly welcome your views via twitter on what they mean for the future of research @ukpenguin



